Chronisch
myeloproliferative
Erkrankungen
Dr. Sormann Siegfried
Polycythemia rubra vera
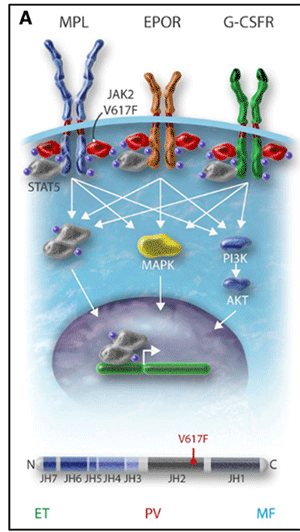
Die Ursache der PRV liegt in einer vermehrten Proliferation aller hämatopoetischen Zellreihen. In nahezu 100% verursacht durch die JAK2-Mutationen V617F zu 96% oder Exon 12 in 4% der Fälle.
JAK2(V617F)homozygous subclones are found both in ET and PV patients, the expansion of a dominant homozygous subclone occurs almost exclusively in PV.
Das verursacht auch die Transformation einer JAK2 positiven ETH in einer PRV.
pruritus after bathing, erythromelalgia, gout, arterial or venous thromboses, hemorrhage, and early satiety due to the presence of splenomegaly
Definition
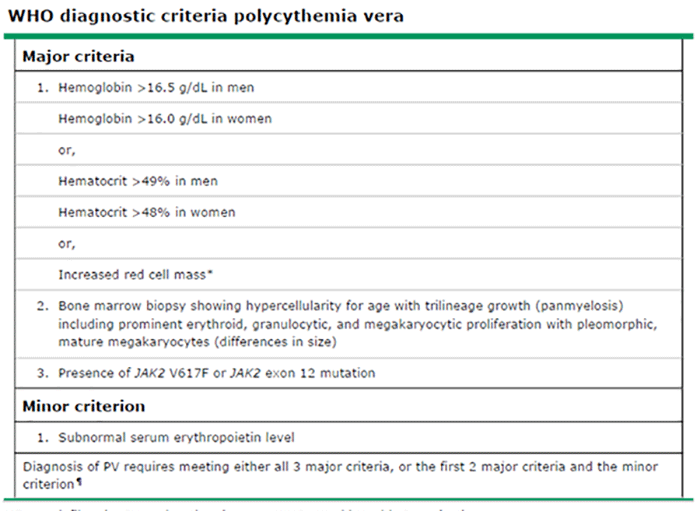 |
| Erythrocytosis is a major diagnostic criterion for PV |
Vorgangsweise
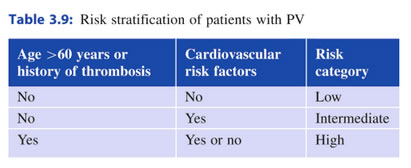
- Anamnese mit Erfassung der cardivaskulären Risikofaktoren - Thrombose
- CRP - Eisenstatus im Labor
- sek vWJ Syndrom bei hoher Thrombozytenzahl
- Oberbauchsono - Milzgröße - PMF - Jamshidi
- bcr-abl Kombinationen kommen vor
- Treibermutationen - JAK2,wenn negativ: Calreticulin,MPL
- Risikoscore
- Knochenmarkpunktion bei jungen Patienten, fortgeschrittener Erkrankung,Splenomegalie (PMF), NGS zur Erfassung modifizierender Mutationen
Pathophysiologie
JAK2 Mutation
 |
Übersicht der Risiken
 |
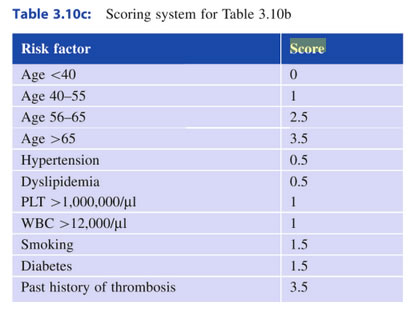
Score
| Alter >70 | 1 Punkt | 84% OAS | 0 Punkt |
| Thrombose | 1 Punkt | 59% OAS | 1 Punkt |
| Leuko >13000 | 1 Punkt | 26% OAS | 2,3 Punkte |
Score 2
| Alter >67 | 5 Punkt | low risk | 0 Punkt |
| Alter 57-67 | 2 Punkt | ||
| Thrombose | 1 Punkt | intermediate risk | 1,2 Punkte |
| Leuko >15000 | 1 Punkt | high risk | 3 Punkte |
Score 3
 |
Score 4
 |
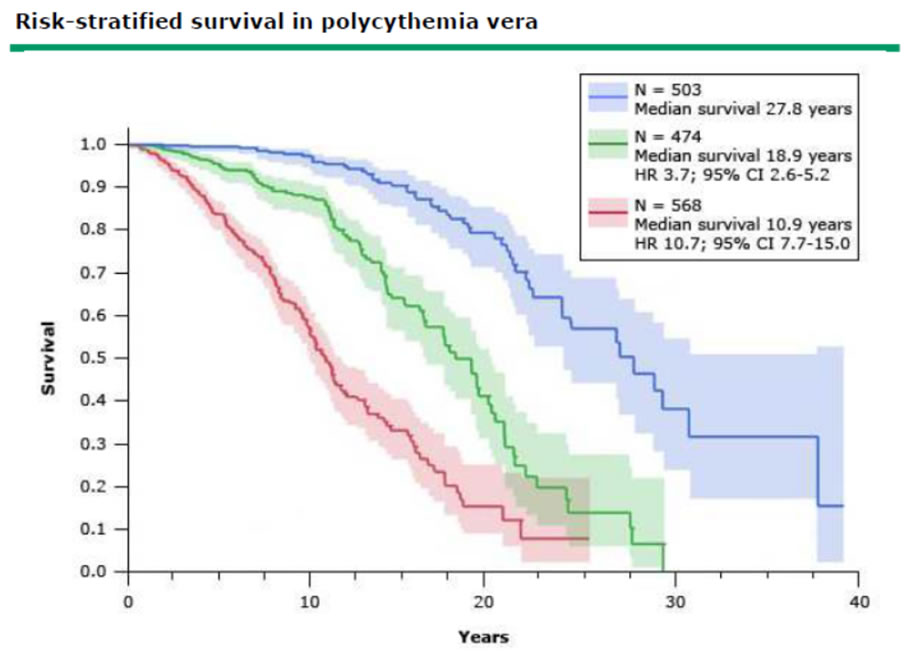
OAS
| OAS | Todesursachen |
| 93% 5 Jahre | 29% Thrombose |
| 72% 10 Jahre | 23% AML,MDS,NHL |
| 46% 20 Jahre | 16% solide Tumore |
| 7% Blutungen | |
| 3% post-PV Myelofiborse |
 |
Therapie
Ziele:
- OAS
- Symptome
- Vorbeugung Thrombose
- Vorbeugung Blutung
Hämatokrit
CYTO-PV trial
As a result of the above studies, we and various authors have suggested that the hematocrit be maintained at less than 45 percent , or at less than 45 percent in men and less than 42 percent in women.
Ziel: Hkt < 45% Männer; <42% Frauen; Thrombo < 400 000
Vorgangsweise
low risk - Phlebotomie + Aspirin
high risk - Phlebotomie + zytoreduktive Therapie + Aspirin
Phlebotomie
A standard one unit phlebotomy (500 mL) should reduce the hematocrit by 3 percentage points in a normal-sized adult (eg, from 46 to 43 percent).
Optimal control is to keep the hematocrit continuously below 45 percent in men and 42 percent in women
Since phlebotomy is effective in controlling PV by producing a state of relative or absolute iron deficiency, iron supplementation should not be given
Aspirin
If not otherwise contraindicated because of a history of major bleeding or intolerance, we suggest that aspirin be given to all patients (Grade 2C). The appropriate dose is 75 to 100 mg/day. Treatment with higher doses should be avoided
A 2013 Cochrane analysis of these two reports [87,90], covering 630 PV patients, concluded that the use of low-dose aspirin (compared with placebo) was associated with a non-significant lower risk of fatal thrombotic events (OR 0.20; 95% CI 0.03-1.14), a non-significant benefit for all-cause mortality (OR 0.46; 95% CI 0.21-1.01), a non-significant increase in minor bleeding (OR 1.85; 95% CI 0.90-3.79), and no significant increase in the risk of major bleeding
Hydroxyurea
primäre Therapie bei älteren Patienten - zytoreduktiv - auch CHR; gute Kontrolle, keine Wirkung auf mutierte HSC; erhöhtes Leukämierisiko nicht sicher; Erstlinie
weiter...
Pegasys
genetisches Ansprechen, Stammzellwirkung,JAK2-VAF Reduktion; LPS; grippale NW; Erstlinie
weiter...
Besemri
genetisches Ansprechen, Stammzellwirkung,JAK2-VAF Reduktion; LPS; grippale NW; pegINF Gabe alle 14 Tage; Erstlinie
weiter...
Jakavi
häufig komplette Remission; JAK2verminderung; Infektneigung; Absetzreaktion; Zweitlinie
weiter...
Thromboreductin
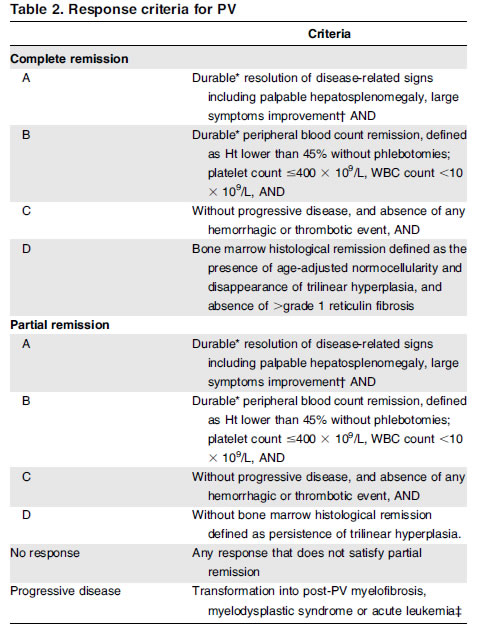
Responsekriterien ELN
Leukämie-Risiko der Therapien

|
